|
Carboxymethylated Beta-(1-3) Glucan
in Skin Care Link to our principals: Mibelle AG Biochemistry,
Switzerland Keywords: CM-Glucan, Yeast
Polysaccharide, UV-A, Oxidative Stress, Skin Protection, Anti-Aging |
![]()
Introduction
Evaluation of
the activity of CM-Glucan by cell culture techniques
Protection
of human skin against oxidative stress induced by UV-A
Protection of
human skin against detergent treatment
Improvement
of signs of aged and photoaged skin
Conclusion
References
Introduction
Crude extracts from yeast have been used for a long time for cosmetic and
pharmaceutical purposes. These products have been found useful in treating
various diseases and skin conditions [1]. In 1941, investigations of yeast
components led to the discovery of the first defined pharmaceutical yeast
product, Zymosan [2]. Further studies have shown that water insoluble Zymosan
has immune-stimulating activity. This product is a raw cell wall preparation
composed of glucan, other polysaccharides, proteins and lipids. Over the last
two decades, glucan from yeast cell walls has been identified as a single
immunologically active component.
Glucan is a Beta-(1-3)-linked
polyglucose of high molecular weight and belongs to the class of drugs known
today as biological response modifiers. Glucan from baker's yeast is a very
potent stimulator of the immune system by activating macrophages and other
cells. Therefore, glucan preparations have been extensively studied in wound
healing [3], infectiology [4] and oncology [5, 6]. In all these applications,
different Beta-(1-3)-glucan preparations from yeast have been shown to be very
active. Recently, the good tolerability and efficacy of a soluble yeast glucan
has been proven in a phase II study [7]. The application of the soluble glucan
preparation before and after thoracic or abdominal surgery lowered
postoperative infection rates.
Glucan isolated from the cell wall
of baker's yeast (Saccharomyces cerevisiae) is a water insoluble
particulate polymer which is not suitable for topical applications. We have
developed a process to modify Beta-(1-3) glucan from baker's yeast to
carboxymethyl glucan (CM-Glucan), a water soluble product. The
carboxymethylation takes place under specific conditions in a reaction that
yields a product with a substitution degree of 0.75 [8]. The chemical identity
of the structure could be confirmed by 13C-NMR spectroscopy [9]. The
dermatological tolerance of CM-Glucan has been carefully monitored in
dermatologically healthy volunteers using a 2% aqueous solution. The results
prove that this material is neither an irritant/photo-irritant nor a
sensitizer/photo-sensitizer.
Evaluation
of the activity of CM-Glucan by cell culture techniques
Glucan preparations have been
shown to be active at very low concentrations. Wolk et al. found a significant
acceleration in wound healing by topical application of a glucan preparation at
0.01% [3].
We have investigated the activity
of our CM-Glucan on porcine keratinocytes. The addition of the polysaccharide
to the M199 culture medium containing 10% calf serum showed a significant
stimulation of the keratinocytes proliferation. At a concentration of 0.01%,
the relative cell count was increased by more than 40% after 120 hours.
In other experiments, CM-Glucan has
been tested for its ability to protect human skin cells against UV-A
irradiation. In our study, cultures of human keratinocytes were developed from
normal adult skin. The cell cultures from various donors were pretreated with
CM-Glucan for 18 hours at a concentration of 0.01% before they were exposed to
UV-A irradiation (320-450 nm) at a dose rate of 300 Wm-2 for two different time
periods.
The protective effect of this
pretreatment regarding oxidative stress in human skin cells could be
demonstrated by measuring intracellular glutathione and ferritin concentrations
as endpoints [10]. These cell culture experiments showed that CM-Glucan is able
to protect keratinocytes from the depletion of antioxidant molecules.
Protection
of human skin against oxidative stress induced by UV-A
Our in-vitro studies
demonstrated that a pretreatment of the cultures with CM-Glucan had a
substantial protective effects against cell damage induced by UV-A irradiation.
To evaluate the in-vivo efficacy of
the polysaccharide to protect skin against oxidative stress induced by UV-A
irradiation, the non-invasive technique of squalene hydroperoxides
determination was applied [11].
Squalene is one of the main
component of the sebum and is particularly susceptible to photo-oxidation.
Colin et al. [11] showed that even low dose UV-A irradiation of the skin leads
to the formation of squalene hydroperoxides. However, the application of some
free radical scavengers to the skin can protect these lipids against peroxidation.
In our study, three oil-in-water
emulsions containing 0.2%, 0.04% and 0% of CM-Glucan were applied twice daily
on the forearm of ten volunteers. On the fifth day, the pretreated skin and a
non-treated area were exposed to UV-A irradiation (10 J cm-2). Subsequently,
skin lipids were extracted with 1 ml of ethanol from all irradiated areas and
as a control also from non irradiated skin. Squalene and squalene
hydroperoxides were then determined in these extracts by HPLC techniques [11,
12, 13, 14].
A very wide range of squalene
concentrations could be detected in the skin of the different volunteers.
However, in all subjects, the UV-A irradiation lead to a substantial increase
of squalene hydroperoxides concentration. The pretreatment of the skin with the
formulations containing CM-Glucan resulted in a significant reduction of the
peroxidation of squalene (Fig. 1). The protection against oxidative stress with
the formulations containing CM-Glucan can be expressed as percentage inhibition
of peroxidation relative to the placebo preparation. The peroxidation itself is
expressed as the ratio of squalene hydroperoxides to squalene [11]. The
addition of only 0.04% CM-Glucan to the oil-in-water emulsions resulted in a
59% inhibition of the squalene peroxidation. An almost complete protection
(94.9%) against UV-A induced oxidation could be observed in this test with the
product containing 0.2% CM-Glucan [15].
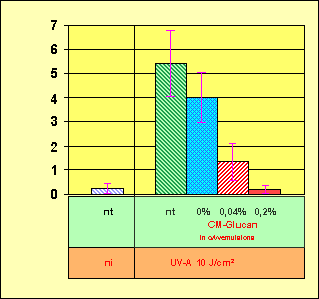
Figure 1. In vivo formation of squalene hydroperoxides
caused by UV-A irradiation. Skin sites of 10 volunteers were pretreated for
five days with oil-in-water (o/w) emulsions containing different concentrations
of carboxymethylated glucan (CM-Glucan). Then the pretreated skin sites and a
non-treated site (nt) were UV-A exposed (10 J cm-2). Squalene hydroperoxides
concentrations were measured in chemiluminescence (CL) units after lipid
extractions from irradiated skin sites and as a control from a non irradiated
(ni) area.
Protection
of human skin against detergent treatment
CM-Glucan was formulated at
different concentrations in a hydrogel and in an oil-in-water emulsion. The
products were applied twice daily to the forearm skin of five volunteers over a
period of 14 days. After this pretreatment, the skin was exposed to sodium
dodecyl sulfate (10%) for two hours.
In the course of the product
application, all formulations enhanced the skin humidity (corneometer units)
compared to the untreated skin (control). The subsequent challenge with sodium
dodecyl sulfate lead to a drastic reduction of skin humidity. However, in skin
pretreated with the products containing CM-Glucan, the reduction of skin
humidity was much less pronounced than in untreated skin. The strength of the
protective effect depends clearly on the concentration of CM-Glucan in the
formulations [16].
The application of the products
over a period of 14 days did not influence the transepidermal water loss of the
skin. The damage of the skin barrier function by the subsequent detergent
challenge lead to an increase of the values for transepidermal water loss. But
a significant dosage dependent protection by the formulations containing
CM-Glucan could be observed compared to placebo [15].
Improvement
of signs of aged and photoaged skin
The aging process of the
skin can be divided into intrinsic changes and extrinsic changes (photoaging)
resulting from chronic exposure to UV-irradiation and other environmental
factors. Intrinsic aging leads only very slowly to obvious changes of the skin.
In contrast, photoaging results in marked changes such as increase of wrinkles,
enhancement of skin roughness, loss of skin firmness and mottled pigmentation.
In general, with advancing age, the
number and activity of immuno-competent cells (Langerhans cells and
keratinocytes) in the epidermis is decreased which makes the skin more
susceptible to environmental hazards. In the dermis, a loss of elastin fibrils
and soluble collagen can be observed. As a result, the skin appears wrinkled
and has lost its elasticity. Preventive and therapeutic approaches to the
problem of photodamaged skin are of considerable importance since photoaging
and also photocarcinogenesis are rapidly increasing in developed nations
throughout the world as a result of increased life span and hence larger
populations of elderly individuals. Topically applied tretinoin at 0.05% has
been successful in the treatment of some of the visible signs of photodamaged
skin after 6 months [17]. However, the marked skin irritation caused by
tretinoin excludes the use of this retinoid in cosmetic applications.
In our investigation, we have
looked for potential "anti-aging" effects of the non irritating
CM-Glucan in a long-term study (28 days) on aged skin. The skin of 10
volunteers (age > 60 years) was treated twice daily around the eyes and on
the forearm with an oil-in-water emulsion containing 0.04% CM-Glucan and the
corresponding placebo product. To simulate photoaging to some extent, the
forearm skin of the volunteers was exposed to a sun simulator at 0.75 MED twice
weekly. At days 1, 14 and 28 the skin firmness and the eye wrinkle depth were
determined.
The irradiation of the forearm skin
twice a week with a sun simulator at 0.75 MED lead to a reduction in skin
firmness after 28 days for untreated skin. The application of the placebo
product improved the skin firmness slightly after 14 and 28 days compared to
untreated skin. The incorporation of 0.04% CM-Glucan into the same emulsion lead
to a statistically significant improvement of skin firmness after 28 days
compared to the placebo product and untreated skin.
The treatment of skin sites around
the eyes with the placebo emulsion lead to a small, statistically not relevant,
deterioration of the wrinkle depth after 14 and 28 days. However, the treatment
with the same emulsion containing 0.04% CM-Glucan markedly improved the skin
condition. After a 28 days treatment with the product containing CM-Glucan, the
eye wrinkle depth could statistically significant be reduced compared to day 1
and compared to placebo treatment.
Conclusion
Our work shows that carboxymethylated Beta-(1-3) glucan (CM-Glucan) from
baker's yeast, with an appropriate degree of substitution, is a new active
component suitable for cosmetic and dermatological applications. CM-Glucan is
water soluble and can therefore readily be used to study efficacy in different
applications. In cell culture experiments, we observed a stimulation of the
viability and the proliferation of keratinocytes at very low concentrations
(0.01%). The pretreatment of human keratinocytes with CM-Glucan rendered them
less sensitive to oxidative stress induced by UV-A irradiation.
Corresponding activities could be
verified in vivo. CM-Glucan enhanced the renewal rate of the stratum corneum
significantly compared to untreated skin [15]. CM-Glucan also offered a
concentration dependent protection against UV-A irradiation. Skin pretreatment
with a formulation containing 0.2% CM-Glucan showed an almost complete
inhibition of lipid peroxidation compared to placebo. Protection against lipid
peroxidation induced by UV-A irradiation is usually only observed by the
application of antioxidants. Colin et al. [11] reported an inhibition of about
90% upon the application of 0.2% D-alpha-tocopherol but only an inhibition of
less than 25% by the application of the cosmetically stable vitamin E acetate
at the same concentration. Since CM-Glucan is neither an antioxidant nor an
iron chelator, it must use mechanisms other than extracellular radical
scavenging or activities related to it. CM-Glucan appears to stimulate cells to
produce endogenous factors which protect the skin against oxidative stress and
other environmental insults.
The pretreatment of skin with
cosmetic formulations containing CM-Glucan at different concentrations showed
substantial protecting effects against skin damage caused by detergent
challenge. In a concentration-dependent manner, CM-Glucan protected the skin
against the decrease of skin humidity and the increase of transepidermal water
loss. To some degree, a second skin effect from the film-forming properties of
the polysaccharide may be present. However, the concentrations used in our
experiments were too low for CM-Glucan to have produced such profound effects
solely through its film-forming behavior.
The treatment of aged skin (age of
volunteers > 60 years) with an oil-in-water emulsion containing 0.04%
CM-Glucan clearly improved skin firmness and eye wrinkle depth even compared to
the placebo control and not only to untreated skin. A statistically significant
effect of CM-Glucan could be observed after 4 weeks of treatment indicating
that overall improvements of photodamaged skin needs some time. In our study, a
marked increase of the effects was observed in the second part of the treatment
period (day 14 to day 28). Therefore, it is reasonable to assume that a
continuation of the treatment might lead to further improvements and additional
effects. However, the exact mode of CM-Glucan activity has not been fully
elucidated and warrants further investigations. The increase of skin firmness
is related to the reduction of wrinkles. It seems that CM-Glucan stimulates the
production of skin factors which improve the elasticity and smoothness of the skin.
In addition, the film-forming properties and the UV-A protecting effects of
CM-Glucan could also contribute to the observed results.
References
1. H?nsel, R. Polysaccharide die immun-stimulierend wirken: Eine
Uebersicht ?ber entsprechende Fertigarzneimittel. Farmaceutisch Tijdschrift
voor Belgie 64, 313-326 (1987)
2. Pillemer, L. and Ecker, E.E.
Anticomplementary factor in fresh yeast. J. Biol. Chem. 137, 139-142 (1941)
3. Wolk, M. and Danon, D. Promotion of wound
healing by yeast glucan evaluated on single animals. Med. Biol. 63, 73-80
(1985)
4. Kokashis, P.L., Williams, D.L., Cook, J.A.
and Di Luzio, N.R. Increased resistance to staphylococcus aureus infection and
enhancement in serum lysozyme activity by glucan. Science 199, 1340-1342 (1978)
5. Di Luzio, N.R., Williams, D.L., McNamee,
R.B., Edwards, B.F. and Kitahama, A. Comparative tumor-inhibitory and
anti-bacterial activity of soluble and particulate glucan. Int. J. Cancer 24,
773-779 (1979)
6. Hofer, M., Pospisil, M., Bohacek, J.,
Pipalova, I. and Sandula, J. Enhancement by carboxymethylglucan of early
cellular damage in 1 Gy-irradiated mice. Folia Biologica Praha 41, 112-117
(1995)
7. Babineau, T. J., Hackford, A., Kenler, A.,
Bistrian, B., Forse, R.A., Fairchild, P. G., Heard, S., Keroack, M., Caushaj,
P. and Benotti, P. A phase II multicenter, double-blind, randomized,
placebo-controlled study of three dosages of an immunomodulator (PGG-Glucan) in
high-risk surgical patients. Arch. Surg. 129, 1204-1210 (1994)
8. Z?lli, F. and Suter, F. Patent pending,
Mibelle AG
9. Z?lli, F. and Saecker, C. CM-Glucan a new
yeast polysaccharide for cosmetic use. Cosmetic and Toiletries Manufacture
Worldwide. 131-136 (1994)
10. Z?lli, F., Applegate, L.A., Frenk, E. and
Suter, F. Photoprotective effects of CM-Glucan on cultured human skin cells.
Eurocosmetics 11, 46-50 (1995)
11. Colin, C., Boussouira, B., Bernard, D.,
Moyal, D. and Nguyen, Q.L. Non invasive methods of evaluation of oxidative
stress induced by low doses of ultra violet in humans. IFSCC Congress Venezia A
105, 50-72 (1994)
12. Nissen, H.P. and Kreysel, H.W. The use of
HPLC for the determination of lipids in biological materials. Chromatographia
30, 686-690 (1990)
13. Zhang, J.R., Cazers, A.R., Lutzke, B.S. and
Hall, E.D. HPLC-chemiluminescence and thermospray LC-MS study of hydroperoxides
generated from phosphatidylcholine. Free Rad. Biol. Med. 18, 1-10 (1995)
14. Holley, A.E. and Slater, T.F. Measurement
of lipid hydroperoxides in normal human blood-plasma using
HPLC-chemiluminescence linked to a diodearray detector for measuring conjugated
dienes. Free Radical Research Communications 15, 51-63 (1991)
15. Z?lli, F., Suter, F., Biltz, H., Nissen,
H.P. and Birman, M. Carboxymethylated ?-(1-3)-Glucan. Cosmetics and Toiletries
111, 91-98 (1996)
16. Z?lli, F., Suter, F., Blitz, H. and Nissen,
H.P. CM-Glucan: A biological response modifier from baker’s yeast for skin
care. S?FW-Journal 123, 535-541 (1997)
17. Gilchrest, B.A. Retinoids and photodamage.
Br. J. Dermatol. 127, 14-20 (1992)
For
Further information feel free to contact us:
A. Baz Marketing Ltd.
P.O.Box 4925
Caesarea 38900
Israel
Tel.: +972 - 4 - 6363841
Fax: +972 - 4 - 6360841
